Tag: temperature conditions
Stability Testing Requirements: Temperature and Time Conditions for Drug Products
By Lindsey Smith On 24 Feb, 2026 Comments (15)
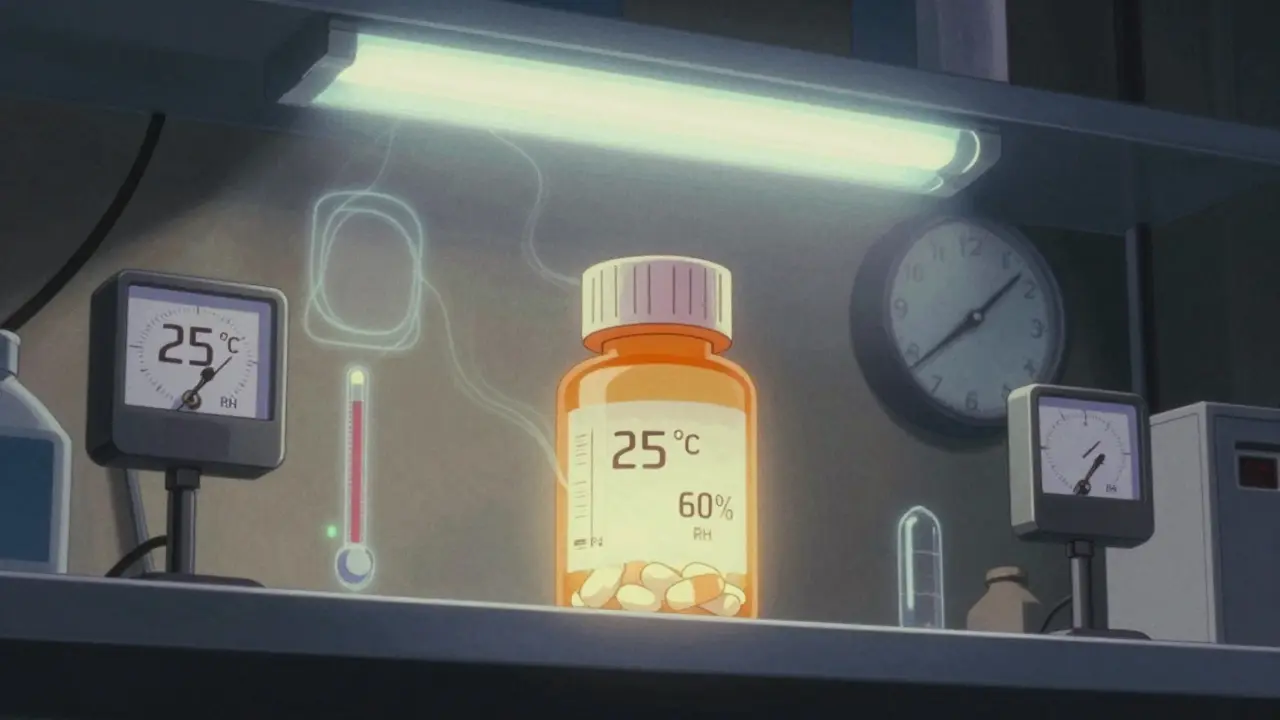
Stability testing for pharmaceuticals requires strict temperature and time conditions to ensure drug safety and efficacy. ICH Q1A(R2) sets global standards for long-term, accelerated, and intermediate testing, with precise humidity and temperature controls. Failure to comply can lead to recalls, delays, or regulatory action.
View More