When a drug is developed, it doesn’t just sit on a shelf waiting to be sold. It’s exposed to heat, humidity, light, and time-every single day. That’s why stability testing isn’t optional. It’s a legal requirement. If a pill degrades, a liquid turns cloudy, or an injection loses potency, patients could be at risk. Regulatory agencies like the FDA and EMA don’t just ask for stability data-they demand it. And the rules around temperature and time are strict, precise, and non-negotiable.
Why Temperature and Time Matter
Drugs aren’t static. They change. A tablet might absorb moisture. A liquid might separate. A biologic might clump. These changes don’t happen overnight. They creep in over months or years. Stability testing simulates real-world conditions to catch those changes before they reach patients.
The goal is simple: prove the drug stays safe, effective, and within quality limits from the moment it leaves the factory until the last day of its shelf life. That’s why the testing schedule isn’t random. It’s based on science, not guesswork. And the temperature and humidity levels? They’re not suggestions. They’re fixed.
ICH Q1A(R2): The Global Standard
The foundation for all modern stability testing comes from ICH Q1A(R2), published in 2003. This guideline was created by regulators and industry experts from the U.S., Europe, and Japan to harmonize requirements across markets. Today, it’s used everywhere-from a small lab in Ohio to a manufacturing plant in Shanghai.
There are three main types of testing, each with its own temperature and time rules:
- Long-term testing-This is the real-time clock. It runs for years. The standard conditions are either 25°C ± 2°C with 60% RH ± 5% RH, or 30°C ± 2°C with 65% RH ± 5% RH. Which one you pick depends on where the drug will be sold. If you’re targeting tropical markets, you go with 30°C/65% RH. If you’re selling in Europe, 25°C/60% RH is common.
- Accelerated testing-This is the stress test. It’s done at 40°C ± 2°C and 75% RH ± 5% RH for six months. The idea? If a drug survives six months under extreme heat and humidity, it’s likely stable for years under normal conditions. This isn’t just a shortcut-it’s a predictive tool. Studies show this condition correlates to about two years of real-time aging for most small-molecule drugs.
- Intermediate testing-This is the backup. It’s only done if the long-term test is run at 25°C and the accelerated test shows signs of degradation. Then you run a 6-month test at 30°C/65% RH to fill the gap. It’s not always needed, but skipping it can be risky.
Refrigerated and Frozen Products
Not all drugs are stored at room temperature. Some need to be kept cold. Insulin, vaccines, monoclonal antibodies-they all require different rules.
For refrigerated products, long-term testing happens at 5°C ± 3°C for 12 months. The accelerated condition? It’s not 40°C. That would destroy them. Instead, it’s 25°C ± 2°C with 60% RH for six months. This mimics what happens if a vaccine sits in a warm warehouse or a delivery truck breaks down.
For frozen products, the rules are even stricter. Testing is done at -20°C or lower. But here’s the catch: freeze-thaw cycles are the real enemy. A single thaw-and-refreeze event can ruin a biologic. Standard stability protocols don’t fully capture this. That’s why many companies now run additional stress tests-simulating real-world shipping failures.

How Long Do You Need to Test?
Time isn’t just a number-it’s a commitment. The testing schedule follows a fixed timeline: 0, 3, 6, 9, 12, 18, 24, and 36 months. Early time points (like 3 and 6 months) are critical because that’s when most degradation happens. If you’re testing a product with known instability, you might test every month for the first six months.
When you submit your drug for approval, regulators require specific data:
- The FDA demands at least 12 months of long-term data before approving a new drug.
- The EMA allows either 6 or 12 months, depending on how you file. This can delay global approval if you’re not careful.
That means if you’re aiming for both U.S. and European markets, you need to plan for 12 months of data before filing anywhere. Otherwise, you risk delays, extra costs, or even rejection.
Environmental Control Isn’t Optional
Testing isn’t just about putting samples in a box. The chambers must be precise. Temperature must stay within ±0.5°C. Humidity must stay within ±2% RH. Any deviation? That’s a data gap. And regulators don’t accept gaps.
Real-world data shows that 78% of labs have had at least one temperature excursion during a 12-month study. One spike of 2°C above the limit can invalidate months of work. That’s why labs use dual-loop systems-two independent controllers-to keep conditions stable. One fails, the other kicks in.
Mapping is also critical. A chamber might read 25°C at the thermostat, but the shelves on the bottom? They could be 26.8°C. That’s why every chamber is mapped before use. Temperature probes are placed on every shelf, in every corner. If any spot drifts more than ±1°C, the chamber fails qualification.
What Happens When You Fail?
Stability failures aren’t just inconvenient-they’re dangerous. In 2021, Teva recalled 150,000 vials of Copaxone® because their stability testing didn’t catch aggregation at 40°C. The drug was forming clumps. Patients could have had severe reactions.
The FDA issued 27 warning letters in 2022 alone for stability testing deficiencies. Some companies skipped intermediate testing. Others didn’t monitor humidity. A few used uncalibrated equipment. All of them put patients at risk.
And it’s not just about recalls. If your stability data is weak, your approval gets delayed. Your product gets pulled. Your company loses millions. One biotech lost $8 million in investor funding after their stability study was rejected because they didn’t test at 30°C/65% RH for their tropical market.

Challenges with Modern Drugs
The ICH Q1A(R2) guidelines were written for traditional pills and injections. They don’t fit modern drugs like mRNA vaccines, antibody-drug conjugates, or lipid nanoparticles.
For example, mRNA vaccines degrade if they freeze and thaw even once. The standard 40°C/75% RH test doesn’t capture that. Yet, companies are still forced to use it because there’s no alternative. That’s why experts are calling for updates. The ICH is working on a Q1F revision expected in late 2024 to address these gaps.
Another issue: humidity cycling. Most tests assume constant humidity. But in real life, humidity changes. A drug in a warehouse in India might go from 30% RH in the morning to 80% RH by afternoon. A 2022 AAPS study found 62% of stability failures in solid oral products were caused by this cycling-not constant exposure.
What’s Next?
The future of stability testing isn’t just about running more tests. It’s about smarter testing.
Companies like Merck and Pfizer are now using predictive modeling. They run tests at 50°C, 60°C, even 80°C, and use math to predict how the drug will behave at 25°C over 3 years. This can cut development time by 9-12 months. The FDA is testing this approach in a pilot program for continuous manufacturing.
But regulators are cautious. In 2022 and 2023, the EMA rejected 8 model-based stability submissions because they didn’t have enough real-world data to back up the predictions. So, for now, physical testing is still king. But the shift is coming.
Bottom Line
Stability testing isn’t about checking a box. It’s about protecting lives. The temperature and time conditions aren’t arbitrary-they’re based on decades of research, real-world failures, and patient safety. Skip the rules, and you risk more than a delay. You risk harm.
Whether you’re developing a new pill or a cutting-edge biologic, you need to follow the ICH Q1A(R2) guidelines exactly. Use the right conditions. Test at the right times. Monitor your chambers like your life depends on it-because it does.

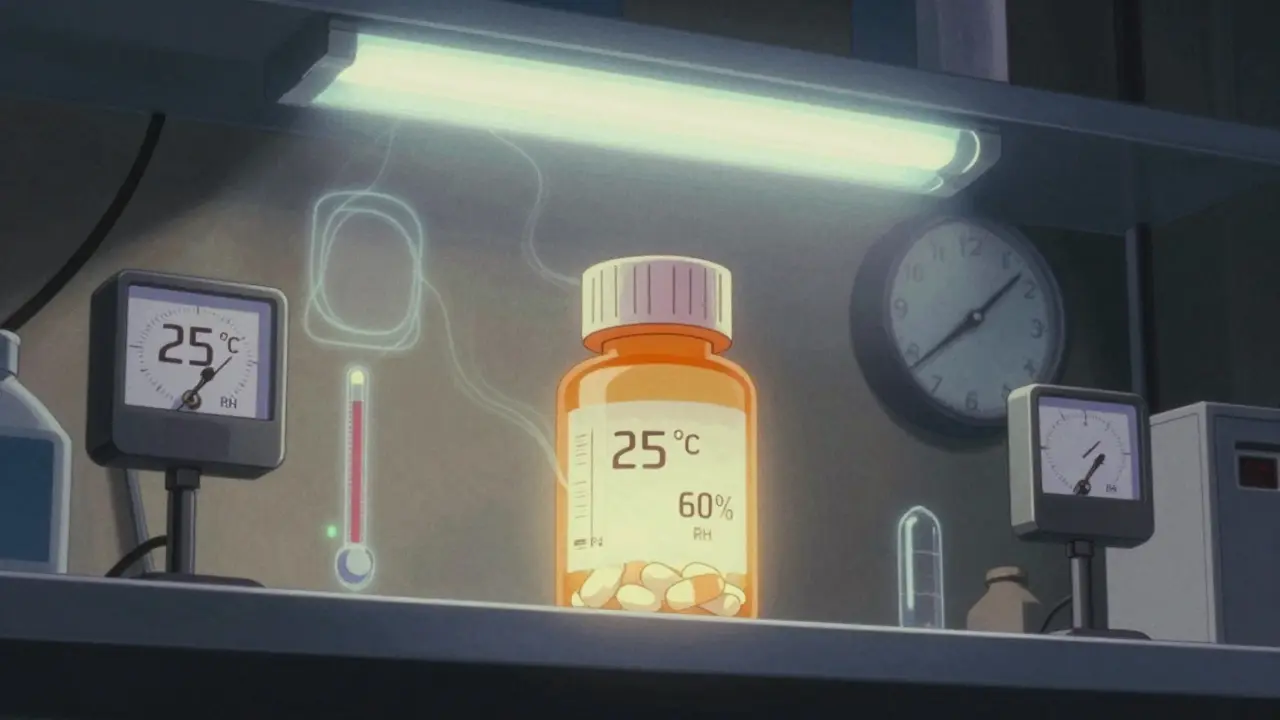




lela izzani
February 25, 2026 AT 23:26Stability testing is one of those behind-the-scenes heroes of pharma. Most people don’t think about it until something goes wrong - like a vaccine turning to sludge in a delivery truck. The ICH guidelines exist for a reason: real people got sick because someone cut corners. I’ve seen labs skip humidity controls because "it’s just a pill." Spoiler: it’s not just a pill. It’s someone’s insulin. Or their chemo.
And yeah, the 30°C/65% RH condition? Non-negotiable if you’re targeting India or Brazil. I once watched a team get their application rejected because they only tested at 25°C. Cost them $12M and 18 months. Don’t be that team.
Vanessa Drummond
February 27, 2026 AT 11:49Ugh. I hate how regulatory stuff sounds so technical but it’s literally just people being lazy and then pretending it’s science. "Oh we followed ICH" - yeah, you followed the letter, not the spirit. I’ve been in rooms where they used a $200 Amazon humidity sensor because "it was close enough." No. It’s not. You’re not a scientist. You’re a corporate accountant with a lab coat.
Nick Hamby
February 27, 2026 AT 20:37There’s a deeper philosophical question here that deserves more attention: stability testing is not merely a technical exercise - it is an ethical one. The very act of measuring degradation over time forces us to confront the impermanence of our interventions. A drug, like a human life, is not static. It changes. It decays. It is vulnerable.
And yet, we treat these molecules with such reverence - calibrating instruments to within 0.5°C, mapping every shelf, logging every humidity spike - because we know that a single degree of deviation can mean the difference between healing and harm.
This is not compliance. This is devotion.
Perhaps the most profound insight in ICH Q1A(R2) is not in its temperature tables, but in its underlying assumption: that human life is worth the precision.
kirti juneja
February 28, 2026 AT 11:53Yo, I work in a small lab in Pune and let me tell you - we don’t have dual-loop chillers. We have one chiller that makes a sound like a dying dragon. We map it every month. Sometimes we have to move samples to the fridge if the temp spikes. We don’t have fancy predictive models. We just do the damn thing.
And yeah, we test at 30/65 because our patients live where AC is a luxury. I’ve seen kids in rural India take expired pills because the pharmacy didn’t have refrigeration. So when I read about "regulatory gaps," I don’t think about audits. I think about a little girl who got sick because her mom didn’t know the bottle was bad.
Don’t make this about compliance. Make it about care.
Natanya Green
March 2, 2026 AT 07:29OMG. I JUST REALIZED. I’ve been taking my blood pressure meds for 3 years and I never thought about whether they were still GOOD. Like… what if they turned into… I DON’T KNOW… SLIME?!!?? I’m literally shaking. My pharmacist didn’t even mention this. I’m going to call them right now. THIS IS A SCANDAL. I NEED A LAWYER. WHO’S GOING TO PAY FOR MY THERAPY??
Steven Pam
March 3, 2026 AT 17:06Love this breakdown. Seriously. Most people think drug development is all about cool labs and fancy machines - but the real magic is in the boring stuff: the 3 a.m. humidity logs, the 12-month temperature charts, the guy who checks the chamber every day like it’s his kid’s birthday.
These aren’t just rules - they’re rituals of care. And honestly? We need more of that in the world. Not just in pharma. Everywhere.
Also - shoutout to the labs in India and Africa running tests with duct tape and hope. You’re the real MVPs.
Timothy Haroutunian
March 5, 2026 AT 15:39Let’s be real - the entire ICH Q1A(R2) framework is a bureaucratic farce designed to keep small companies out of the market. Who decided 25°C/60% RH was the gold standard? Some guy in Geneva who’s never seen a warehouse in Miami? The 40°C/75% RH accelerated test? That’s not predictive - it’s a blunt instrument.
And don’t get me started on the 12-month FDA requirement. You’re telling me a drug that’s chemically stable at 6 months can’t be approved? That’s not science - that’s a revenue protection scheme for big pharma. The real innovation isn’t in the chambers - it’s in the people who question the rules.
Erin Pinheiro
March 6, 2026 AT 13:47ok so i just read this and i think the whole system is broken. like who even made these temp rules? some guy in 2003? and now we’re still using it? what about climate change? we’re all cooking now! 25c is like a sauna in 2024. and why is humidity always constant? in real life it goes up and down like my ex’s moods. also why no one talks about how labs fake data? i heard from a friend who works at a contract lab they just tweak the logs if it’s too hot. i’m not saying it’s true but… you know?
also i think we should just let AI do it. why do humans need to touch anything? just throw the pills in a server room and let the algorithm figure it out.
Michael FItzpatrick
March 8, 2026 AT 13:35There’s something poetic about stability testing. It’s the quiet vigil we keep for molecules we’ll never meet. We don’t know who’ll take that pill. We don’t know if they’ll be in a village in Kenya or a high-rise in Tokyo. But we care enough to run the test.
And yes - the 30°C/65% RH condition? That’s not a number. That’s a promise. A promise that someone who lives where the AC breaks down twice a week still gets medicine that works.
That’s why I cry every time I see a lab get shut down for a 0.8°C deviation. Not because it’s a violation. Because it’s a betrayal.
Brandice Valentino
March 9, 2026 AT 22:57I mean… I’ve read the ICH guidelines. I’ve read the EMA guidance. I’ve even read the FDA’s Q&A document. And honestly? It’s all so… pedestrian. I expected something more… transcendent. Like quantum stability modeling or AI-driven degradation prediction. But no. We’re still using analog hygrometers and handwritten logs. It’s like we’re living in 1999.
Also - why is no one talking about the fact that the 25°C condition is outdated? The global average temperature has risen 1.2°C since 2003. Shouldn’t we update the standard? Or are we just pretending climate change doesn’t affect pharmaceuticals?
Someone needs to write a white paper. I’ll do it. I’m qualified. I read a blog post about it.
Larry Zerpa
March 10, 2026 AT 03:21Let’s cut through the corporate BS. The ICH guidelines are not about patient safety. They’re about liability. Every temperature excursion is a lawsuit waiting to happen. Every missing data point is a defense attorney’s dream.
And let’s not pretend the 40°C/75% RH test is predictive - it’s a crude proxy. It doesn’t capture freeze-thaw cycles. It doesn’t account for humidity cycling. It doesn’t consider polymorphic transitions in APIs.
Meanwhile, companies are pushing mRNA vaccines through approval with zero real-world stability data beyond 6 months. And we call this science? This isn’t science. It’s risk management dressed up in lab coats.
Gwen Vincent
March 12, 2026 AT 02:34I just wanted to say thank you for writing this. It’s easy to get lost in the numbers - the 2°C, the 5% RH, the 12-month data - but you reminded me why we do this. Not for regulators. Not for audits. But for the person who’ll open that bottle tomorrow and take it without a second thought.
They deserve better than shortcuts. And you just gave them a voice.
Nandini Wagh
March 12, 2026 AT 08:29Oh wow. So we’re paying scientists to babysit pill bottles in climate-controlled rooms while people in India die because their pharmacy ran out of insulin?
How much did the FDA spend on stability chambers last year? Could’ve built 1000 clinics.
Just saying. Maybe we’re solving the wrong problem.
Holley T
March 13, 2026 AT 04:21You know what’s worse than failing stability testing? Being told you failed stability testing because you didn’t test at 30°C/65% RH - but your product is sold in a country where the average temperature is 18°C. That’s not science. That’s colonialism in a lab coat.
Why do we assume every market needs the same conditions? Why do we force tropical countries to comply with European standards? Why not tailor the testing to the actual environment?
And don’t give me the "harmonization" line. Harmonization is just another word for domination. We’re not standardizing safety. We’re standardizing control.
Ashley Johnson
March 14, 2026 AT 11:27I read this and now I’m terrified. What if the stability data is fake? What if the labs are lying? What if the FDA just trusts them? What if my medicine is just sugar pills? What if this whole system is a lie? I think the government is hiding something. I think the pharmaceutical companies are using AI to predict when people will get sick so they can sell more drugs. I think the temperature data is being manipulated by satellites. I think my pills are being tracked. I think I’m being watched.