The FDA Orange Book is the official publication listing approved drug products with therapeutic equivalence evaluations. Meanwhile, the Purple Book is the counterpart for licensed biological products, focusing on biosimilarity and interchangeability. Using these tools isn't just for regulatory experts; it's a practical way to perform drug safety monitoring and ensure a medication switch won't lead to an adverse reaction.
Why These Books Matter for Patient Safety
Not all generic drugs are created equal. Some are therapeutically equivalent, meaning they work the same way and have the same safety profile. Others aren't. If you swap a brand-name drug for a generic that isn't a perfect match, you could face diminished efficacy or unexpected side effects.
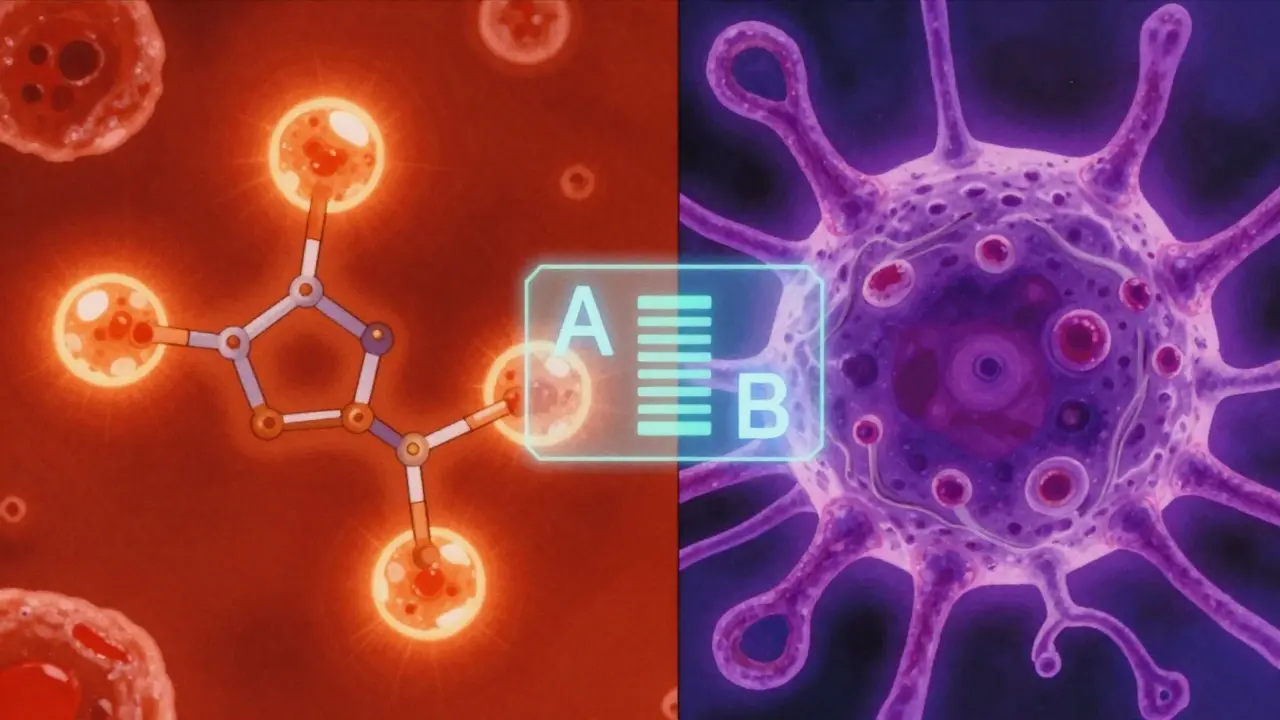
The Orange Book helps you spot these differences using specific codes. For example, an "A" code tells you the product is therapeutically equivalent to the reference drug. If you see a "B" code, the drug is not considered therapeutically equivalent, even if it contains the same active ingredient. This distinction is a critical safety guardrail, especially for medications with a narrow therapeutic index where a tiny change in dosage or absorption can be dangerous.
Biologics are more complex. Unlike simple chemical drugs, biologics are made from living cells. Because they are so complex, they can't be "generic" in the traditional sense; they are "biosimilars." The Purple Book allows you to see if a biologic has been proven to have no clinically meaningful differences in safety compared to the original reference product. Without this check, switching biologics is a gamble with patient stability.
Navigating the Orange Book for Safety Red Flags
If you want to check if a drug was pulled from the market due to a safety crisis, you shouldn't just search the main list. You need to head straight to the "Discontinued Section."
The FDA explicitly flags products that were withdrawn for safety or effectiveness reasons. As of late 2023, over 120 products are listed here. To find these, follow these steps:
- Access the Orange Book searchable database on the FDA website.
- Use the "Drug Product Selection" filter.
- Set the filter to "Discontinued Drug Products."
- Select "Safety" as the reason category.
A hospital pharmacist once shared a story about catching a dangerous substitution because the Orange Book showed a generic had been withdrawn for safety issues, while the brand-name version was still active. This is exactly why these databases are a primary line of defense in clinical settings.
Using the Purple Book to Evaluate Biologics
The Purple Book operates differently because biologics aren't just "equivalent"-they are either biosimilar or interchangeable. A product labeled as "interchangeable" has met a higher safety bar, meaning the risk of switching back and forth between the biosimilar and the reference product is no greater than using the reference product alone.
To evaluate a biologic's safety:
- Search by the reference product (the original brand) first.
- Look at the "Biosimilarity or Interchangeability" column.
- A "Yes" in this column indicates the product has met the FDA's rigorous comparability requirements.
One of the best features of the Purple Book is how it groups all biosimilars under their reference product. This lets you see a side-by-side safety comparison without jumping between different documents. However, remember that a biosimilarity a-okay doesn't guarantee an identical reaction in every single patient; it just means the profile is clinically comparable.
Comparing the Two Resources
While both books aim for safety, they serve different purposes based on the type of medicine you're investigating. The Orange Book is about chemical "sameness," whereas the Purple Book is about biological "comparability."
| Feature | Orange Book | Purple Book |
|---|---|---|
| Primary Focus | Small molecule (chemical) drugs | Biological products (biologics) |
| Safety Indicator | Therapeutic Equivalence Codes (A/B) | Biosimilarity/Interchangeability Status |
| Withdrawal Data | Explicit "Discontinued Section" for safety | Reference product grouping for comparison |
| Update Cycle | Typically every 30 days | Typically every 60 days |
| Safety Goal | Verification of generic substitution | Verification of biological comparability |
One major difference is the update speed. The Orange Book usually updates faster, while the Purple Book can have a longer lag. This means if a safety alert just broke today, the Orange Book might reflect it sooner than the Purple Book.

Crucial Limitations and How to Fill the Gaps
Here is the most important thing to remember: neither the Orange Book nor the Purple Book provides a full list of side effects or real-time adverse event reports. They tell you if a drug is approved and equivalent, but they don't tell you if a specific patient is having a bad reaction.
To get a complete safety picture, you need to use these books in tandem with other tools:
- FAERS (FDA Adverse Event Reporting System): Use this for detailed data on actual adverse events reported by doctors and patients.
- FDA MedWatch: Check here for the latest safety alerts and recalls that might not have hit the "Books" yet.
- FDA Safety Communications: Essential for post-marketing updates that occur between the books' update cycles.
If you only rely on the Purple Book, you might miss a recent post-marketing safety update. Cross-referencing is the only way to ensure total patient safety.
Practical Tips for Healthcare Providers
If you're using these tools daily, efficiency is key. Pharmacists typically spend 2-5 minutes on an Orange Book check and 5-10 minutes on a Purple Book check due to the complexity of biologics. To speed things up, focus on the "Reference Product Exclusivity" column in the Purple Book. This tells you when safety data exclusivity has expired, which often signals that more comprehensive comparative safety data is available.
For those struggling with complex safety scenarios-like drugs with a narrow therapeutic index-the FDA offers free 90-minute training webinars. These are highly recommended because interpreting "B" codes in high-risk medications requires more nuance than a simple search can provide.







julya tassi
April 21, 2026 AT 08:32This is such a helpful breakdown! I've always been a bit nervous about switching to generics without knowing the 'why' behind the safety checks. :)
Valorie Darling
April 21, 2026 AT 15:45imagine thinking we need a whole guide for this lol just ask your pharmacist instead of spending an hour digging through a govt database
William Young
April 21, 2026 AT 22:37It's worth noting that while the tools are great, the human element of pharmacy is still the biggest safety net. These databases are supplements, not replacements for clinical judgment.
Lesley Wimbush
April 23, 2026 AT 12:52Honestly, it's just so quaint that some people still struggle with these basics. I've known about the A and B codes since my first year of residency, but I suppose not everyone has that level of academic rigor.
It's absolutely tragic that patients have to navigate this themselves because the system is so flawed, but here we are, pretending a basic PDF search is a revolutionary act of safety monitoring. I'll be the one to say it: the Purple Book is an absolute nightmare to navigate if you aren't properly trained, and pretending it's a simple 'yes/no' check is just misleading. Let's not pretend we're all experts just because we can use a search bar on a government website. It's frankly embarrassing that this needs to be explained in such basic terms for a general audience, but I suppose we must cast a wide net for the sake of public health, however tedious it may be for those of us who actually understand the biochemistry involved.
Tanya Rogers
April 24, 2026 AT 05:24The premise that accessibility equals understanding is a fallacy. Mere access to the Orange Book does not grant one the pharmacological insight to interpret the systemic implications of a 'B' rating in the context of narrow therapeutic index drugs.
Brigid Prosser
April 24, 2026 AT 23:11Stop overcomplicating it with the fancy talk! The bottom line is that these tools are absolute goldmines for anyone who actually cares about their health and doesn't want to be a guinea pig for some cut-rate generic. Get in there, do the digging, and don't let the bureaucracy scare you off!
Charlotte Boychuk
April 25, 2026 AT 03:33It's a bit of a wild ride trying to figure out biologics, isn't it? Glad there's a way to cut through the noise and see what's actually interchangeable without having to guess.
Cynthia Didion
April 25, 2026 AT 11:03FDA standards are the gold standard worldwide. Period.